
Establishing an FDA office in the Middle East/North Africa: An Abraham Accords Initiative
“Change is not required. Survival is not mandatory.”
–W. Edwards Deming
Introduction
On December 2013, the U.S. Food and Drug Administration (FDA) closed its office inside the American embassy in Amman, Jordan. According to an agency spokesperson, “This is a transfer of agency personnel rather than a reduction.” No reasons were given for this action.[1] The Jordanian office of the FDA opened in 2011. According to a 2012 report to Congress, FDA employees from the office were leading training efforts in Saudi Arabia in good clinical practices for the Middle East and North Africa (MENA) drug regulators, among other activities.
It’s time to revisit this decision. Relative to United States’ interest, FDA needs a MENA office. There are two main reasons, both reinforced by the recent Abraham Accords[2] (currently signed by the United States, Israel, the United Arab Emirates, Bahrain, and Morocco). The first reason is to enhance American pharmaceutical supply chain security via “nearshoring.” The second reason is to both expedite and lower the cost to the American market of innovative medical technologies (biopharmaceuticals, medical devices, diagnostics, nutraceuticals, etc.) by expanding the capabilities of the MENA region’s Abraham Accords nations to conduct cutting-edge research, development, and regulatory review. Let’s look at each opportunity.
I. Nearshoring
Traditionally, nearshoring is where a business moves its operations to a nearby country from one of greater distance, for basic logistical benefits.[3] An FDA office within the Abraham Accords boundaries addresses another benefit of nearshoring—a dependable supply chain. Specifically, nearshoring will address American pharmaceutical supply chain security by:
(1) Reducing dependency on China for many of the critical medical products that are manufactured, almost exclusively for sale in the United States; and
(2) Diversifying the U.S. supply chain, making it more resilient.
Imported Chinese active pharmaceutical ingredients (APIs) represent around 90% of the American supply for certain life-saving antibiotics, such as cephalosporins, azithromycin, and penicillin.[4]
In 2019, China accounted for 95% of U.S. imports of ibuprofen, 91% of U.S. imports of hydrocortisone, 70% of U.S. imports of acetaminophen, 40–45% of U.S. imports of penicillin, and 40% of U.S. imports of heparin.[5]
Per FDA’s Principal Deputy Commissioner, Dr. Janet Woodcock, “The FDA doesn’t know whether Chinese facilities are actually producing APIs, how much they are producing, or where the APIs they are producing are being distributed worldwide, including in the United States. Similarly, we do not have information that would enable us to assess the resilience of the U.S. manufacturing base, should it be tested by China’s withdrawal from supplying the U.S. market.”[6]
The COVID-19 pandemic together with increasing political friction between the United States and China makes this an urgent American national security issue. Earlier this year, due to a pandemic-driven shutdown of Shanghai, a GE Healthcare plant was forced to interrupt its manufacture of iodinated contrast media used in many imaging exams. As a result, GE Healthcare’s contrast media were added to the FDA drug shortage list on May 9, 2022.[7] This shortage forced the delay of many essential medical diagnostic tests. A tertiary manufacturing facility for contrast media in a nation friendly to the United States, and with the technological capability and sophisticated regulatory advice and oversight, would play an important role in securing American pharmaceutical security.
FDA should assess the urgency of the problem and whether Abraham Accords partner nations would help develop solutions. To those ends, FDA should, as a strategic national security objective, develop a list of essential medical technologies that are primarily manufactured (API, finished form, etc.) in China. This would help to solve the problem of medical product shortages before they happen.
The United States wants to reduce its dependance on medical products produced in countries that could weaponize this dependence. This doesn’t exclusively mean more manufacturing within our own borders, but by other countries with whom we have closer ties and greater trust. Such a “near-shoring” strategy reflects the spirit of the Abraham Accords and its member nations.
II. Expediting the Development and Introduction of Life-Saving Therapies for American Patients
The development of new medical technologies is a global proposition. Competition incentivizes investment, drives success, lowers prices, and increases patient access.
An Abraham Accords-based FDA presence in the region will foster deeper and more regular collaboration between regional MENA regulatory agencies and FDA, which would advance safety and efficacy standards of food and medical products manufactured within individual nations and across the region.
This regulatory collaboration will also provide technical assistance to local and regional developers and manufacturers, so that more locally developed products can obtain FDA approval for sales in the United States. This will also result (with enhanced FDA technical assistance) in more locally based clinical trials and other technical endeavors (i.e., Chemistry Manufacturing and Control/CMC protocols, etc.) crucial to navigating medical product development and, ultimately, the FDA review process.
A precedent for this effort is the “Expanding Medical Partnerships with Israel to Lessen Dependence on China Act,”[8] included as part of the National Defense Authorization Act for Fiscal Year 2021. This Act authorized $4,000,000 yearly for collaborative life sciences research. Future legislation could increase funding, expand regulatory oversight, and incentivize private sector investment.
Such an initiative recognizes the significant public and private investment) of Israel and the United Arab Emirates in advanced technological and intellectual infrastructure.
In this respect, the role of an FDA MENA office will parallel the objectives of FDA’s European office,[9] which are (per the FDA website):
- Fostering collaboration with the FDA’s regulatory counterparts to identify opportunities for regulatory alignment, to the extent possible; identifying and analyzing key legislative and policy developments in Europe and then advocating for the FDA’s positions in consultation with Centers and Offices;
- Promoting the FDA’s safety and efficacy standards and the advancement of FDA’s international objectives that contribute to improvements in global public health; and
- Supporting the U.S. Interagency Mission to the European Union to ensure the coordination of U.S. government activities that are related to FDA equities.
A further strategic parallel is FDA’s view that working closely with European regulatory authorities will help to identify and facilitate early FDA engagement with global regulatory partners in emerging areas spanning a range of FDA-regulated products. The aim is to exchange information, discuss rapidly evolving scientific evidence, and promote convergence in policy formulation, where possible. In consultation with relevant FDA Centers and Offices, such on-the-ground assistance supports FDA’s broader objectives to advance and realize the potential of digital health technologies, technology and data modernization, and innovation in plant and animal biotechnology, as well as combatting illicit trade in FDA-regulated products. All these timely and important opportunities exist within the boundaries of the current (and hopefully expanding) Abraham Accords nations.
One goal of these collaborative partnerships should be to develop the groundwork for a Pharmaceutical Mutual Recognition Agreement (MRA).[10] Under such an agreement, the United States and the member nations of the Abraham Accords would rely on information from inspections of drug manufacturing facilities conducted within each other’s border, freeing up valuable regulatory resources. An FDA office in the region would serve a critical role in conducting capability assessments and facilitating other steps necessary to establish and implement MRA processes. This includes conducting an evaluation of the applicable local regulatory framework to determine whether a given counterpart MENA agency has the capability, capacity, and procedures to carry out routine Good Manufacturing Practice (GMP) surveillance inspections that meet FDA requirements.
III. Abraham Accords Nations: State of Play and Future Opportunities for Mutual Benefit Through Enhanced Regulatory Fraternity
A series of information-gathering conversations with senior diplomats, health ministry officials, and commercial stakeholders from the Abraham Accords nations offers valuable insights into how each nation could potentially benefit from an on-the-ground FDA presence and how those benefits, in turn, serve the specific interests of the United States.
The United Arab Emirates (UAE)
The UAE has invested billions of dollars to attract, build, and expand both physical and intellectual infrastructure for medical technology development. According to a November 2021 report, the UAE intends to boost investments in the pharmaceutical industry by 20% and widen its export reach in the region.[11] The national policy for medicine provides a framework within which the activities in the pharmaceutical sector can be coordinated and encourages research and development, including operational research, drug development, and clinical research. Part of the plan is to produce more good quality medicines locally and draw more investments into the sector.
Abu Dhabi, as an example, has been steadily and effectively building a reputation as a center of excellence for innovation. The emirate (and federal seat of government) is investing heavily in technology and innovation across all fields of healthcare, pharmaceutical, and biotech companies. In December 2020, the Abu Dhabi Investment Office (ADIO) announced the expansion of its incentive program to include innovation-focused companies in other high-growth areas in demand, including health services, pharma, medtech, and biotech. The program provides $545 million of rebates and grants to foreign companies to support their continued growth in Abu Dhabi.[12]
In 2021, the USTR outlined a set of key achievements made by the UAE in the field of Intellectual Property Protection (IPP) during the past period, leading to its success in coming off the watch list.[13] The report highlighted several measures taken by the UAE to develop its national system for protecting intellectual property rights across various commercial and investment activities, the most important of which are: The Ministry of Health and Prevention resolving concerns with IP protection of pharmaceutical products (Decree 321) and the UAE making progress on longstanding IP enforcement concerns, particularly through increased efforts by Dubai Customs.
According to Ahmed Ali Al Sayegh, UAE Minister of State, “The USTR’s decision is encouraging news. Over the last year, we have worked hard to strengthen the UAE’s IPP framework in many important ways. This decision is an endorsement of our progress. The UAE is committed to implementing robust IPP regulatory standards, including having an infringement and enforcement framework that upholds these standards. We firmly believe that the process of creating and commercializing IP enriches society and drives economic growth. It is, therefore, our intention to build on this positive result and continue to strengthen and reinforce rigorous IPP rights across the UAE.”[14]
Bahrain
Bahrain currently has no FDA-approved sites and very little relationship with FDA. Relative to their own pharmaceutical security, COVID served as a wake-up call to the government, which now recognizes and is taking steps to prioritize the need to protect their own supply chain. While current domestic pharmaceutical manufacturing is minimal, the willingness to invest in this infrastructure is significant and comes from the very highest levels of government. In a post-COVID environment, Bahrain is seeking to become a pharmaceutical manufacturing hub for the Middle East region.[15]
In 2010, the primary responsibility for regulating the health sector was transferred from the Ministry of Health to the National Health Regulatory Authority (NHRA), and in December 2012, the NHRA’s Pharmaceutical Products Regulation Department was established. The department is also responsible for inspecting pharmaceutical factories, examining inventories, and overseeing human resource qualifications.[16]
Bahrain is actively involved with Israel, including partnerships with Sheba Medical Center and serves as the co-chair of the Negev Forum Working Group on Healthcare.[17] Recently, Secretary of State Tony Blinken and the foreign ministers of Bahrain, Egypt, Israel, Morocco, and the United Arab Emirates pledged to strengthen security and economic cooperation and try to bring more countries on board.[18]
Bahrain is excited about the opportunity to work with the FDA to construct manufacturing facilities dedicated to manufacturing specific pharmaceutical products identified as key near-shoring concerns for the U.S. market.[19]
An FDA field office will help them scale up a robust domestic manufacturing industry and offer significant incentive to multinational pharmaceutical companies to invest for the U.S. market. Bahrain has no personal or corporate income tax and has the lowest operating costs in the region.
Opportunities to interact with the FDA to enhance Bahrain’s regulatory expertise are timely and plentiful.
Morocco
Multinational pharmaceutical companies are increasingly seeing Morocco as a top manufacturing destination for the African continent. Firms from Europe, Asia-Pacific, and the United States are investing millions of dollars in bolstering and expanding their drug production facilities in the North African nation thanks to its strategic location, price-competitiveness, and comparatively developed infrastructure.[20]
In May 2019, Mylan Pharmaceuticals opened its first pharmaceutical production plant in Casablanca for Myhep All, a generic medication for the treatment of hepatitis C. Mylan already manufactures several generic molecules for oncology in Morocco.
Beyond building up their manufacturing base, an area of keen interest is advancing Morocco as a preferred site for global clinical trials. Regular and robust interaction with a regional FDA office would be of immediate and intense interest.[21]
The need to access larger numbers of patients and qualified investigators for expanded late phase trials, combined with an already saturated field for patient recruitment, has created demand for a truly global approach to site recruitment. Ease of patient enrollment and high-quality, yet lower-cost clinical resources make the Middle East a truly attractive region for late phase studies.[22]
The Center of Research in Epidemiology Study and Clinical Trials at Mohammed V-Souissi University in Rabat, Morocco has begun building up its skill base with the creation of the Unity of Biostatistics, as well as the formation of a group of clinical trainers and the Workshop of Biostatistics and Methodology for professors in different specialties and began the country’s first Laboratory of Biostatistics and Methodology of Clinical Research.
As a result, Morocco has been able to develop clinical protocols and methodology of drafting of research, conduct statistical analysis as well as epidemiological work of studies, and aid the development of relevant databases. Morocco’s National Drug Quality Control Laboratory has been prequalified by the World Health Organization (WHO) and accredited by the European Directorate for the Quality of Medicines (EDQM), allowing Morocco to be Associate Member of European Pharmacopoeia Commission. Its National Center for Pharmacovigilance is a WHO collaborating organization. Morocco has a Bioequivalence Center in conformity with international standards.
By the end of 2022, the Moroccan Directorate of Medicines and Pharmacy (the FDA equivalent) will no longer be a division of the Ministry of Health, but an independent agency. In conversations with its director, Bouchra Medda, some relevant points in support of an FDA office in the region are:
- Morocco recognizes the danger to its own national security from an over-reliance on Chinese-manufactured pharmaceuticals and vaccines and is developing strategies to address this issue.
- A top strategic goal is to become a hub for quality medicines manufacturing for the African continent.
- They want to attract more clinical trials and clinical research to Morocco.
- They want to develop more FDA-approved manufacturing facilities so they can become a viable alternative for products (innovative and generic) exported to the United States.
But they cannot succeed alone. Morocco needs a trusted partner such as the U.S. Food and Drug Administration.
Israel
Israel, America’s strongest ally in the Middle East, is also a world leader in the field of life sciences research, development, and manufacturing.[23] Israel is renowned for its startup culture and the explosive number of companies conducting research and development of innovative life science products, especially medical devices, and pharmaceuticals. During the teeth of the COVID-19 pandemic, it was Israeli clinical trial data for the Pfizer/BioNTech COVID-19 vaccine that delivered the most comprehensive information, allowing FDA to chart a swifter course towards an Emergency Use Authorization.[24]
Israel is a global leader in Artificial Intelligence (AI)—a key technology for developing new biopharmaceuticals, enhancing GMPs for existing medical technologies, and advancing 21st Century pharmacovigilance. Israel’s Artificial Intelligence ecosystem is undergoing explosive growth, characterized by a major influx of research centers and talent. The Israeli Ministry of Science has designated AI as a “vital area of research,” and in 2019, began offering financial incentives to encourage academic AI research.[25]
Israel is ranked #1 in private R&D expenditure as a percentage of GDP—about 4.3% (as of 2016), of which 84% comes from the private sector—the highest among OECD countries, reflecting the prospering private sector innovation ecosystem. Israel is also ranked #2 at the Innovation Index of the Global Financial Forum, which includes parameters such as scientific research institutions, business sector R&D expenditures, academy–industry cooperation, scientists and engineers pool, and the number of patents in ratio to the size of the population.[26]
Cumulative Number of Active Life Science Companies (2011–2020)[27]
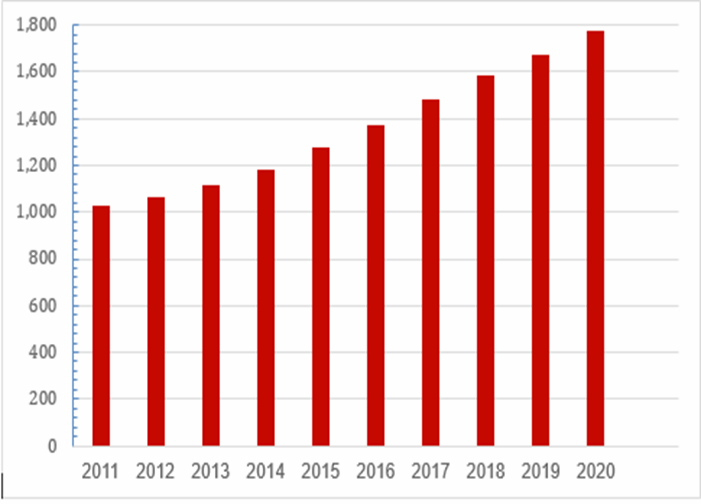
In 2013, Israel and the European Union signed an agreement on GMPs. If the Israeli Ministry of Health approves manufacturing facilities for drugs and devices, the EU recognizes these sites as acceptable for the European market.[28] FDA does not have a similar agreement with Israel for reciprocity. While there are some FDA-approved sites in Israel, a similar agreement would be very helpful. In the meantime, FDA needs to send someone from the United States to physically inspect a facility to approve. An FDA field office staffed with real technical experts, not liaison officers, would help streamline this process.
Final Thoughts and Next Steps
Opening an FDA office isn’t simple or inexpensive. As discussions of this initiative move forward, some key questions to address and answer are:
- How should this office be staffed? What kinds of regulatory expertise are most important to the Abraham Accords nations and how will they best address the needs of the United States (pharmaceutical security, commercial and academic partnerships, scientific and regulatory advice, etc.)?
- Where should the FDA office be located? Where will the agency (and the American public) get the biggest bang for the regulatory buck?
- What should the goals and objectives of this office be? Who should be consulted in the development of these plans? What are the metrics for success?
- How much will it cost FDA and where will the additional dollars come from?
- What should the timeline look like for advancing this agenda?
- Who are the stakeholders who should be consulted in this process?
Difficult? Surely. But, as Winston Churchill reminds us, “A pessimist sees the difficulty in every opportunity; an optimist sees the opportunity in every difficulty.”
[1] https://www.outsourcing-pharma.com/Article/2013/09/25/US-FDA-to-close-Middle-East-office-in-December
[2] https://www.state.gov/the-abraham-accords/
[3] https://www.forbes.com/sites/forbestechcouncil/2021/05/27/beyond-outsourcing-how-to-get-started-with-nearshoring/?sh=1b0d76e01799
[4] https://www.cnbc.com/2022/05/27/india-needs-to-fill-china-gaps-to-become-the-pharmacy-of-the-world.html
[5] https://www.politico.com/news/2019/12/20/policymakers-worry-china-drug-exports-088126
[6] https://www.fiercepharma.com/manufacturing/how-much-does-u-s-rely-china-for-drugs-fda-simply-doesn-t-know
[7] https://www.ge.com/news/press-releases/ge-healthcare-update-on-iodinated-contrast-media
[8] https://www.govinfo.gov/app/details/BILLS-116hr6829ih
[9] https://www.fda.gov/about-fda/office-global-operations/europe-office
[10] https://www.fda.gov/international-programs/international-arrangements/mutual-recognition-agreement-mra
[11] https://www.zawya.com/en/business/uae-to-raise-investments-in-pharmaceutical-industry-by-20-cwzdg8v7
[12] https://www.reuters.com/article/emirates-investment-adio-idAFD5N2HK00T
[13] https://www.khaleejtimes.com/uae/us-removes-uae-from-intellectual-property-protection-watch-list
[14] Ibid.
[15] Discussions with officials from the Bahrain Ministry of Health
[16] https://oxfordbusinessgroup.com/analysis/bahrains-pharmaceuticals-industry-takes-new-strides
[17] https://www.axios.com/2022/06/29/negev-forum-israel-bahrain-uae-us
[18] https://www.axios.com/2022/03/28/negev-summit-isreal-uae-egypt-morocco-blinken
[19] Discussions with officials from the Embassy of Bahrain
[20] https://pharmaboardroom.com/articles/morocco-becoming-a-pharma-manufacturing-hub-for-africa/
[21] Discussions with officials from the Embassy of Morocco
[22] https://www.parexel.com/application/files/8015/5682/5392/WP-PERIMIDEAST-FEB14.pdf
[23] https://www.iati.co.il/files/files/IATI%20Israeli%20Life%20Sciences%20Industry%202019%20Report.pdf
[24] https://www.reuters.com/world/middle-east/israel-present-covid-19-booster-shot-data-fda-experts-2021-09-05/
[25] https://www.forbes.com/sites/gilpress/2020/03/03/israel-applies-ai-to-healthcare-building-on-leadership-in-cybersecurity-digital-medical-records/?sh=7dbcfde7fa1d
[26] https://www.weforum.org/reports/the-global-competitiveness-report-2016-2017-1/
[27] https://www.iati.co.il/resource_item.php?ID=568
[28] https://www.ema.europa.eu/en/partners-networks/international-activities/bilateral-interactions-non-eu-regulators/israel
Update Magazine
Spring 2023

 PETER J. PITTS is President of the Center for Medicine in the Public Interest and a Visiting Professor at the University of Paris Medical School. He is a former FDA Associate Commissioner and member of the United States Senior Executive Service.
PETER J. PITTS is President of the Center for Medicine in the Public Interest and a Visiting Professor at the University of Paris Medical School. He is a former FDA Associate Commissioner and member of the United States Senior Executive Service.





