The Hundred Years War: The Fight Against Weight Loss Fraud
For decades, weight loss scams have plagued consumers wanting to manage their weight. The consumer losses have been in the billions, not to mention the frustration that so-called miracles cause when failure ensues, as it inevitably does. Over nearly a hundred years, the Federal Trade Commission (FTC) has battled against purveyors of bogus weight loss remedies. Historically, the agency has relied on a three-prong approach emphasizing enforcement, media responsibility, and consumer education. Through enforcement, it has returned millions of dollars to consumers. However, the future is not clear. Weight loss scams persist, and the agency’s priorities in this area are anything but clear.
Introduction
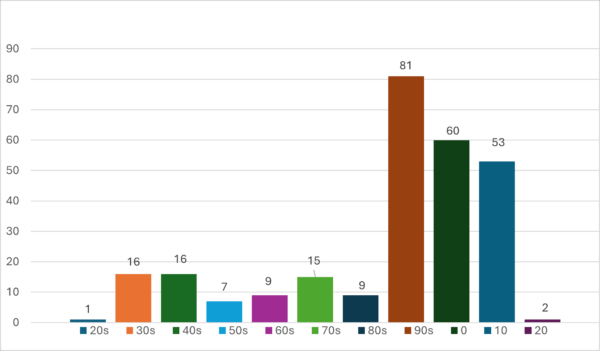
For nearly 100 years, the Federal Trade Commission (FTC) has been battling weight loss fraud.[1] Since filing its first case in 1926, it has initiated at least 268 enforcement actions against companies and individuals who falsely claimed that their products or services would successfully remove unwanted weight and fat (Fig. 1). The FTC’s targets have ranged from fly-by-night charlatans to well-established corporate citizens. Targeted products and services include drugs, dietary supplements, devices, and weight loss programs. The FTC has returned millions of dollars to defrauded consumers. This article reviews the FTC’s enforcement and educational initiatives targeting weight loss fraud. Unfortunately, while battles have been won, the problem persists.
On July 13, 1926, the FTC filed its complaint against McGowan Laboratories, Inc.[2] The complaint also named Womanhood Publishing Corporation, the publisher of a monthly magazine called True Romances. Advertisements for the product, marketed under the name McGowan’s Reducine, claimed that the cream would “[literally dissolve excess fat], leaving the figure slim and properly rounded, giving the lithe grace to the body that every man and woman desires,” and “applying [the product] and patting it gently onto the parts of the human body which the purchaser desires to slenderize, will thereby reduce any and every part of the body so treated, no matter how much or how little excess flesh there might be, quickly, surely, and permanently.”[3] The FTC alleged that the claims were false and therefore constituted an unfair method of competition. While the claims at issue in McGowan would be familiar today, this case is unique because, in addition to proceeding against the marketer of the product, the FTC named the publisher of the magazine that ran the ad as a respondent in the case. While decades later the FTC would turn its attention to the role of the media in facilitating weight loss fraud, this is the first and only time to date that the FTC has filed a false advertising case against a legitimate publisher. Neither McGowan nor Womanhood Publishing contested liability, and the Commission entered an order prohibiting the companies from disseminating false weight loss claims in the future. It was only the beginning.
The Scope of Weight Loss Fraud
Few other consumer frauds have been as pervasive over so long a period as weight loss fraud. The FTC conducted surveys measuring the level of mass-market consumer frauds in 2005, 2011, and 2017.[4] The 2017 survey found that weight loss fraud was the most common fraud reported, representing approximately 6.5 million consumers.[5] These results are consistent with the results of mass-fraud surveys the FTC conducted in 2005[6] and 2011.[7]
Consumer injury from weight loss fraud is not limited to financial loss. Some fraudulently promoted products have posed serious health risks for consumers. For example, phenylpropanolamine, which was the active ingredient in the product the FTC challenged in the Porter and Dietch[8] (discussed below) and Taleigh Corporation[9] cases, has been linked to hemorrhagic stroke.[10] Ephedra, an ingredient challenged in the FTC’s case against USA Pharmacal Sales,[11] among others, has been associated with heart attacks, strokes, and seizures.[12] Not all cases involve ingredients with the potential for direct serious physical injury, but even when there is no reason to suspect that the ingredients may be dangerous, consumers using deceptively marketed weight loss products may be forgoing proven remedies to their detriment. Obesity and overweight[13] are serious health conditions.[14] Consumers who turn to unproven weight loss remedies are putting their health at risk.
It remains a matter of speculation as to why some consumers are susceptible to unrealistic and fraudulent weight loss claims. Societal pressures to lose weight may play a role. Overweight and obese consumers may be vulnerable to such claims given past unsuccessful attempts to lose weight and keep it off. Another factor may be the lack of satisfactory FDA-approved drug products for weight control. In that regard, the emergence of GLP-1 products may have a significant effect on consumer behavior, at least in the short term.[15] The availability of FDA-approved GLP-1 products may divert consumers from questionable substitutes. However, not every consumer will be able to afford a prescription drug product. Moreover, many of the traditional compounds such as garcinia cambogia, CLA, and Green Tea Extract are still being marketed.[16] Additionally, there are new products being marketed as GLP-1 enhancers that deserve a closer look.
Federal Regulations
In general, except for prescription weight loss drugs,[17] the FTC exercises primary regulatory authority over the advertising of weight loss products.[18] The U.S. Food and Drug Administration (FDA), for its part, regulates the labeling of food, nonprescription drugs, and devices and the advertising and labeling of prescription weight loss products. Under the Federal Food, Drug, and Cosmetic Act (FDCA), the regulatory scheme depends on how the product is classified. Prior to 1994, nutritional supplements intended to cause weight loss were regulated as drugs.[19] In 1991, the FDA completed a ten year-long review of 113 ingredients and determined that 111 of those ingredients lacked sufficient evidence to demonstrate effectiveness for weight loss.[20] Following publication of the final monograph, nutritional supplements intended to cause weight loss would have required premarket FDA approval. The monograph would have allowed an efficient mechanism to pull fraudulent weight loss products off the market, potentially saving consumers millions of dollars over the last two decades. Unfortunately, the Dietary Supplement Health and Education Act of 1994 (DSHEA)[21] rendered the FDA’s weight loss monograph moot. Under DSHEA, nutritional supplements marketed for weight loss or weight maintenance do not require premarket approval, whereas the same products intended for the treatment of obesity (which is considered to be a disease) are deemed to be drugs and require FDA premarket approval.[22] This is a dubious distinction for two reasons. First, the line between overweight and obesity is arbitrary. While it may make sense as a medical matter to distinguish between persons who are overweight and those who are obese, it is not clear why that distinction should be relevant to advertising. It does not make sense to treat the same compound as a drug or a dietary supplement depending on whether the ad depicts an obese person in it. Second, the target audience for weight loss products is essentially the same regardless of how the marketing is crafted. While the FDCA does require that claims for nutritional products be substantiated,[23] and the FDA has adopted the FTC’s competent and reliable scientific evidence analytical framework,[24] the FDA has not, to date, enforced the substantiation requirement.[25]
As a result, enforcement at the federal level has been left primarily to the FTC. The FTC Act prohibits unfair methods of competition and unfair or deceptive acts or practices in or affecting commerce[26] and the false advertisement of food, drugs, devices, services, and cosmetics.[27] Under the FTC Act, a weight loss advertisement is deceptive if any material claims[28] in the ad are false or unsubstantiated.[29] An advertisement can also be deceptive or unfair if the advertiser fails to disclose material facts, such as the need to diet and exercise when using the product.[30]
Weight loss efficacy claims must be substantiated by competent and reliable scientific evidence, which in the case of ingestible products, topicals, and devices, requires at least one well-controlled human clinical study.[31] Such studies should be randomized, double-blinded (unless the marketer can demonstrate that blinding cannot be effectively implemented given the nature of the intervention), conducted by persons qualified by training and experience to conduct such studies, measure appropriate endpoints, and yield statistically significant results based on a comparison of the treatment and control treatments.[32] The quantity and quality of competent and reliable scientific evidence required for programs and services will vary depending on the program or service and what experts in the field would consider to be adequate substantiation.[33] For example, weight loss clinics will often rely on their records of patient outcomes for substantiation.[34]
FTC remedies for violations of the FTC Act, FTC’s Trade Regulation Rules, or other statutes enforced by the FTC include injunctions and cease and desist orders prohibiting future deceptive practices, compliance and reporting requirements, notifications to past purchasers, and in limited circumstances, civil penalties and consumer restitution. In 2021, the U.S. Supreme Court held that the FTC lacked authority to seek consumer restitution or disgorgement under Section 13(b) of the FTC Act.[35] Prior to this decision, the FTC routinely sought consumer redress in its weight loss cases filed in federal courts.[36] Currently, the FTC may still seek consumer restitution in certain limited situations, such as rule violations, or in administrative settlements.[37] In addition to prohibiting misrepresentations, FTC orders typically require that advertisers possess a specific level of substantiation for future claims. Currently, that standard is at least one well-controlled human clinical trial that is (1) randomized, double-blind, and placebo-controlled; and (2) conducted by researchers qualified by training and experience to conduct such testing.[38]
Enforcement without the ability to extract a monetary penalty in the form of consumer redress or disgorgement of unjust enrichment has little or no general deterrent effect. Following the Supreme Court’s determination that the FTC did not have authority under Section 13(b) of the FTC Act to obtain either consumer redress or disgorgement from violators, the FTC began focusing its enforcement on rules and statutory provisions that carried civil penalties. As part of this effort, the FTC resurrected its 1983 Notice of Penalty Offenses Concerning Promotion of Products and Plans for Weight Control.[39] This notice, republished in 2022, is based primarily on the Commission’s determination in the Porter & Dietsch case discussed above. It identifies the following claims as deceptive:
- That an appetite suppressant will allow the user to lose weight or fat without dieting or restricting his or her accustomed caloric intake;
- To represent through testimonials that any particular experience with a weight control product or plan reflects the typical or ordinary experience of users of the product or plan, unless the representation is true;
- That use of a weight control product or plan will result in weight loss or a change in body configuration, without possessing and relying upon a reasonable basis for the representation at the time it is disseminated; and
- That scientific evidence supports a statement about a weight control product or plan, without possessing and relying upon competent scientific evidence that fully supports the statement at the time the representation is disseminated.[40]
In theory, this Notice would allow the FTC to obtain a civil penalty against a company that has knowledge that its weight loss advertising violates the prohibited practices identified in the Notice.[41] To establish knowledge, the FTC sends a copy of the Notice to the company. A company that knowingly engages in one of the prohibited practices is subject to a civil penalty of up to $53,088 per violation.[42] To date, the FTC has not invoked this provision.
Enforcement
Historically, the FTC’s battle against weight loss fraud has enjoyed bipartisan support.[43] As noted above, the FTC has initiated hundreds of cases since 1927, and 195 since 1990 (Fig. 1). The types of products and services that have been offered to help consumers lose weight seem unlimited. A partial list includes acai berry, green tea extract, human chorionic gonadotropin, guarana, citrus aurantium, conjugated linoleic acid (CLA), garcinia cambogia, hoodia, green coffee bean, human growth hormone, caffeine-infused shapewear, ab belts, weight loss centers and clinics, and low- and very low-calorie diets. Most of FTC’s enforcement actions have been resolved through settlements, but there are notable exceptions. In litigation, the advertiser has the burden to present the evidence it was relying on to support its claim at the time it was disseminated to the public.[44] The burden then shifts to the FTC to prove that the advertiser’s evidence does not constitute competent and reliable scientific evidence to support the advertiser’s claims. This is usually done through expert testimony.
Figure 1
Number of Weight Loss Enforcement Actions Since 1927 by Decade
The first weight loss case litigated in the modern era was Porter and Dietch.[45] In Porter and Dietch, the FTC initiated enforcement against the manufacturer of the X-11 diet pill (Porter and Dietch), a retailer that disseminated advertisements for X-11 (Pay’n Save Corporation), and the ad agency that produced the deceptive advertisements (Kelly Ketling Furth, Inc.). X-11, a nonprescription drug, purportedly contained 13 active ingredients, including phenylpropanolamine. Advertising claims included “clinically tested ingredient,” “Eat well . . . and lose that fat—without ever missing a meal,” and “No Starvation Dieting—No Strenuous Exercise,” among others. The ads also relied on consumer testimonials, with at least one claiming the user lost 80 pounds. The FTC alleged that ads conveyed that the users could lose weight without dieting and exercise, that substantially all users would lose a significant amount of weight, and that the ads failed to disclose that persons with high blood pressure or heart disease should consult with a doctor and that the testimonials did not represent the typical and ordinary experience of those using the product. The company defended, arguing that phenylpropanolamine hydrochloride was a recognized appetite suppressant and that they were selling a “plan,” not just a pill. The Commission rejected these arguments and found that there was no reasonable basis to support Porter & Dietch’s efficacy claims. In probably the most notable quote in any weight loss advertising case, the Commission noted: “[D]ieting is the conventional method of losing weight. But it is equally obvious that many people who need or want to lose weight regard dieting as bitter medicine. To these corpulent consumers the promises of weight loss without dieting are the Siren call, and advertising that heralds unrestrained consumption while muting the inevitable need for temperance if not abstinence simply does not pass muster. Where dieting is required, there is simply no substitute for clear and conspicuous disclosure that dieting is required.”[46]
A more recent case involved green coffee bean extract. In FTC v. NPB Advertising, Inc., the defendants formed a company to market green coffee bean extract as a weight loss aid after watching a video clip of Dr. Oz describing a clinical study purporting to show that participants taking green bean coffee extract lost 17 pounds in 22 weeks without dieting.[47] The defendants’ sales exceeded $30 million. Defendants’ marketing included claims that the product, marketed under the name of Pure Green Coffee, caused “tremendous weight loss,” including 17 pounds in 22 weeks, 17 pounds in 12 weeks, and 28 pounds in 9 weeks. Based on expert testimony, the court found that the FTC was entitled to summary judgment on its allegation that defendants’ weight loss claims were not supported by competent and reliable scientific evidence. The Court rejected defendants’ reliance on news articles, blog entries, and manufacturers’ brochures as substantiation. The defendants also relied on eight studies and a meta-analysis to support their claims. The court rejected studies that were not conducted on human subjects because there was no showing that the results from the studies could be extrapolated to humans. Defendants primarily relied on a cross-over study of green coffee extract involving 16 subjects separated into three treatment groups—high dose, low dose, and no dose—with a two-week washout period. In this study, subjects lost more weight when not consuming green coffee bean extract than when consuming the test ingredients. For example, the greatest weight loss occurred during the first washout period in a group that had not used the green coffee bean extract. There were numerous other problems with the reliability of the study, including issues with the blinding. Although this study was published, it was subsequently retracted.[48] Defendants’ other studies did not fare better. The Court rejected other studies because they did not involve weight loss as an end point, did not use green coffee extract, or had other methodological flaws, including failure to provide sufficient information to evaluate the reliability of the study, failure to report the drop-out rates, failure to document participants’ caloric intake and exercise, insufficient study length, and failure to describe the blinding and randomization process. Defendants’ proffered meta-analysis[49] did not help defendants given that it concluded that “the effect of green-coffee extract on weight loss, if [any], [i]s minimal.”[50]
Sweeps
Sweeps are one of the FTC’s most effective enforcement tools. In a sweep, instead of announcing a single case, multiple cases are included in a single announcement. There is usually a common theme for the cases. This approach generally results in greater publicity, thereby enhancing consumer education and deterrence. For example, in 1997, the FTC announced Operation Waistline.[51] As part of this initiative, the FTC brought enforcement actions against seven firms for deceptive weight loss claims. According to the FTC, the goal was to “alert consumers to misleading and deceptive weight loss claims, to steer them to accurate information about healthy weight loss, and to continue to bring law enforcement actions against those in the industry who violate the law.”[52] The challenged products and services included cellulose/ox bile, devices, low-calorie diets/very low-calorie diets (lcd/vlcd), and nutritional supplements. In total, the companies agreed to pay $787,500 in consumer redress or disgorgement. As part of Operation Waistline, the FTC also sent advisory letters to more than 100 publishers that ran the weight loss advertisements challenged in the FTC’s complaints, requesting that they improve their advertising review efforts. The FTC noted in its press release that U.S. consumers spend an estimated $6 billion dollars a year on misrepresented weight loss products and services.
In 2011, the FTC filed actions against 10 operators using fake news sites to market acai berry for weight loss.[53] Defendants masqueraded their advertising to look like legitimate news to trick consumers into purchasing acai berry weight loss products. Some advertisements claimed that users could lose up to 25 pounds in four weeks. The complaints sought injunctive relief prohibiting false and deceptive weight loss claims and other deceptive practices and restitution for consumers, who paid between $70 and $100 for the deceptively marketed products. These actions resulted in the recovery of over $5.5 million dollars for consumer redress and nearly $33 million in judgments.
In 2014, the FTC announced enforcement actions against four companies as part of “Operation Failed Resolution.”[54] The companies named were Sensa Products, LLC; HCG Diet Direct, LLC; L’Occitane; and Leanspa, LLC. In the case of Sensa, the company agreed to pay $26.5 million to settle the FTC charges. According to the FTC complaint, Sensa misrepresented that the product caused an average loss of 30 pounds without dieting or exercise.[55] Company sales exceeded $364 million. Sensa consisted of a powder that consumers were instructed to sprinkle on their food to curb their appetite. In December 2014, the FTC sent refund checks to Sensa consumers totaling $26,023,329.00.
Interagency and International Cooperation
The FTC’s jurisdiction extends to all forms of marketing originating in the United States—whether directed to U.S. or foreign consumers—as well as marketing originating outside of the United States that results in sales to U.S. consumers. To date, international cooperation has been limited to Canada and Mexico. In 2005, in what is the largest enforcement effort against weight loss fraud, the United States (FTC and FDA), Mexico, and Canada jointly announced enforcement actions against nearly 730 companies.[56] The compliance actions included prosecutions, recalls, seizures, import refusals, warnings and other enforcement programs against allegedly false and misleading weight loss advertising and labeling. At the same time, the regulatory agencies provided comprehensive education and public outreach programs to help consumers, industry, and advertisers identify deceptive and misleading claims.
Media Screening
Publishers are the conduit between the charlatans and their victims, and as such, they have played a pivotal role in the perpetuation of weight loss fraud over the last century. In 2000, the FTC initiated a campaign to encourage publishers to assume a larger role in protecting consumers from weight loss hucksters who were abusing publishers’ credibility to defraud consumers.
Absent a special dispensation from Congress,[57] the media are not immune from liability under the FTC Act. There is no exception in either Section 5 or 12 of the FTC Act for publishers, and it is well-established that deceptive speech is not constitutionally protected.[58] Accordingly, publishers may be liable for knowingly disseminating false weight loss advertisements. The initial stage of the FTC media responsibility initiative was to document the scope of the problem. In September 2002, the FTC released a report, “Weight Loss Advertising: Analysis of Current Trends,” analyzing 300 weight loss ads that ran in 2001.[59] The report concluded that almost 40% of the ads made at least one claim that was obviously false, and another 15% made claims that were likely to be false or unsubstantiated.[60] The report also compared the 2001 sample with a sample of ads from 1991 and concluded that the 2001 ads were more likely to contain obviously false claims than the 1992 ads.[61] In other words, things were getting worse, not better. In November 2002, the FTC hosted a workshop on Deception in Weight Loss Advertising.[62] The workshop was comprised of three panels, representing science, industry, and the media. The science panel considered several common weight loss claims and concluded that the following claims made for nonprescription drugs, dietary supplements, creams, wraps, devices, and patches were not scientifically feasible at the current time:
- Consumers who use the advertised product can lose two pounds or more per week (over four or more weeks) without reducing caloric intake and/or increasing their physical activity.
- Consumers who use the advertised product can lose substantial weight while still enjoying unlimited amounts of high calorie foods.
- The advertised product will cause permanent weight loss (even when the user stops using the product).
- The advertised product will cause substantial weight loss through the blockage of absorption of fat or calories.
- Consumers who use the advertised product (without medical supervision) can safely lose more than three pounds per week for a period of more than four weeks.
- Users can lose substantial weight through the use of the advertised product that is worn on the body or rubbed into the skin.
- The advertised product will cause substantial weight loss for all users.
- Consumers who use the advertised product can lose weight only from those parts of the body where they wish to lose weight.[63]
In early 2003, FTC Chairman Timothy Muris addressed the Cable Television Advertising Bureau and urged executives to “Do the Right Thing” and take affirmative action to protect consumers from weight loss fraud. Specifically, Chairman Muris encouraged the media to screen out weight loss advertising that made clearly implausible and patently false claims.[64]
Later in 2003, the FTC announced its Red Flag initiative. The initiative was aimed at encouraging media to voluntarily screen out weight loss ads that contained facially false claims. To assist, staff provided a list of false claims as determined in its 2002 workshop and detailed above.[65] The Red Flag campaign was rebranded in 2014 as “Gut Check.” The message, however, remained the same.[66]
In late 2004, the FTC filed law enforcement actions against six firms making fraudulent weight loss claims as part of its “Big Fat Lie” initiative.[67] All cases involved at least one “Red Flag” claim for a variety of products, including pills, powders, green tea, topical gels, and diet patches. The ads ran in publications such as Cosmopolitan, Woman’s Own, Complete Woman, USA Weekend, Dallas Morning News, San Francisco Chronicle, Cleveland Plain Dealer, and Albuquerque Journal, and in Spanish-language publications such as TeleRevista Magazine. The FTC staff followed up with reminder letters to media outlets that ran the challenged advertisements.
As a follow-up to the FTC’s Red Flag initiative, in April 2005, the FTC released the results of a survey of weight loss advertisements that appeared in 2004.[68] The report concluded based on its survey of weight loss product advertisements in print, television, and radio that there had been a significant decline in the incidence of Red Flag weight loss claims between 2001 and 2004.[69] The report noted that staff could not determine with certainty whether the observed results were the result of the FTC’s initiative and, in any event, the decline in Red Flag claims did not necessarily mean that there was a decline in deceptive weight loss claims more generally.[70]
Consumer Education
Consumer education is an important component of the FTC’s efforts to protect consumers from weight loss fraud. As part of that effort, in October 1997, the FTC hosted a conference to explore how the government, industry, and consumer groups could work together to provide consumers with the information necessary to evaluate weight loss adverting.[71] The conference, hosted by FTC, the American Society of Clinical Nutrition, the National Institute of Diabetes and Digestive and Kidney Diseases, and the Centers for Disease Control and Prevention, was designed to consider such topics as what motivates consumers to lose weight, what information consumers need to evaluate products and programs, and the best ways to get consumers the information they need to make an informed decision. As a follow up to the conference, in 1999, a coalition of 41 organizations and individuals, including industry groups, public advocacy organizations, government agencies, scientific associations, and academia, released voluntary guidelines designed to help providers of weight loss products and services give consumers clear and easily understood information on costs, health complications, staff qualifications, maintaining weight loss, and how to improve their chances of long-term success.[72] The FTC also released a new consumer education brochure called Setting Goals for Weight Loss and a video public service announcement.
The FTC’s current consumer education on weight loss scams can be found by going to the FTC’s website or going directly to ftc.gov/weightloss. The website provides useful information on how to spot false promises and recognize common techniques used to induce consumers to purchase products they wouldn’t otherwise buy, as well as tips for purchasing weight loss equipment and joining exercise facilities. The site also provides additional resources and information on how to report weight loss scams.
Conclusion
Over the last century, weight loss scams have been one of the most prevalent consumer frauds. The FTC has a long history of combating weight loss scams, reaching its peak from 1990 to 2020. However, recent FTC enforcement has been almost non-existent. Although the FTC republished its Notice of Penalty Offenses in 2022, publishing rules is not the same as enforcing those rules. Unfortunately, given the inadequacy of the current regulatory tools, including the lack of premarket approval, FTC’s restricted ability to recover monetary redress for consumers, and limited liability for online service platforms, effective deterrence may be difficult to obtain. Moreover, based on the FTC’s case selection under both the Biden and Trump Administrations, there has been a shift away from health product cases generally to an emphasis on budget-balancing frauds, e.g., business opportunities, where the FTC’s ability is obtain civil penalties and consumer restitution are well established.
It is likely that the FTC’s current lack of interest in enforcement has been noticed by marketers of weight loss products and services. Whether the lack of a realistic enforcement threat results in an increase in deceptive and fraudulent weight loss claims to the detriment of consumers remains to be seen.
[1] The Federal Trade Commission Act. The FTC Act was approved on September 26, 1914. See 15 U.S.C. §§ 41-58, as amended (1914).
[2] McGowan Laboratories, Inc., 11 F.T.C. 125 (1927).
[3] Id. at 126.
[4] Press Release, Federal Trade Commission, FTC Releases Results of 2017 Mass-Market Consumer Fraud Survey (Oct. 31, 2019). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2019/10/ftc-releases-results-2017-mass-market-consumer-fraud-survey.
[5] Federal Trade Commission, Mass-Market Consumer Fraud in the United States: A 2017 Update (2019), p 31. Retrieved from https://www.ftc.gov/system/files/documents/reports/mass-market-consumer-fraud-united-states-2017-update/p105502massmarketconsumerfraud2017report.pdf.
[6] Federal Trade Commission, Consumer Fraud in the United States: The Second FTC Survey (2007), p. S-1. According to the survey, an estimated 4.8 million U.S. consumers purchased fraudulent weight loss products. Retrieved from https://www.ftc.gov/sites/default/files/documents/reports/consumer-fraud-united-states-second-federal-trade-commission-survey-staff-report-federal-trade/fraud.pdf.
[7] Federal Trade Commission, FTC Consumer Fraud in the United States (2013): The Third FTC Survey, p. I. Retrieved from https://www.ftc.gov/sites/default/files/documents/reports/consumer-fraud-united-states-2011-third-ftc-survey/130419fraudsurvey_0.pdf.
[8] Porter & Dietsch, Inc., 90 F.T.C. 770 (1977), modified, 605 F.2d 294 (7th Cir. 1979), modified, 95 F.T.C. 806 (1980).
[9] Taleigh Corp., 119 F.T.C. 835 (1995).
[10] Food and Drug Administration, Science Background – Safey of Phenylpropanolamine (2000). Retrieved from https://www.fda.gov/drugs/information-drug-class/science-background-safety-phenylpropanolamine.
[11] Press Release, Federal Trade Commission, FTC Charges Direct Marketers of Ephedra Weight Loss Products With Making Deceptive Efficacy and Safey Claims (July 1, 2003). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2003/07/ftc-charges-direct-marketers-ephedra-weight-loss-products-making-deceptive-efficacy-safety-claims.
[12] Agency for Healthcare Research and Quality, U.S. Dep’t of Health and Human Servs., Ephedra and Ephedrine for Weight Loss and Athletic Performance: Clinical Efficacy and Side Effects, (2003), p. 223.
[13] The Centers for Disease Control and Prevention defines overweight for persons over 20 years of age as a BMI between 25 and 30 and obesity is defined as a BMI over 30.
[14] National Institute of Diabetes and Digestive and Kidney Diseases, Health Risks of Overweight and Obesity (2025). Retrieved from https://www.niddk.nih.gov/health-information/weight-management/adult-overweight-obesity/health-risks.
[15] Weight loss miracles seem to come and go. In terms of its utility as a long-term weight loss solution, it is too early to know what the long-term effect of GLP-1 products will be. Moreover, as demonstrated by the FTC’s case against Southern Health Solutions, the emergence of GLP-1 products will not necessarily eliminate the need for enforcement. Southern Health Solutions, Inc., File No. 232-3040 (consent order) (Dec. 3, 2025).
[16] It is noted that some of the worst claims that appeared in earlier decades no longer appear on some of the major online platforms.
[17] The regulation of the labeling and promotion of prescription drugs is outside the scope of this article.
[18] Federal Trade Commission and Food and Drug Administration, Working Agreement between FTC and FDA, 4 Trade Reg. Rep. (CCH) ¶ 9,850.01 (1971) (MOU).
[19] The FDA Act defines “drug,” in part, to mean any article “intended to affect the structure or any function of the body of man.” Federal Food, Drug, and Cosmetic Act (FDCA), Section, § 201(g)(1), 21 U.S.C. § 321(g)(1)(2025). An article marketed for the purpose of causing cause weight loss is intended to affect the structure and function of the body. See Nutrilab, Inc. v. Schweiker, 713 F.2d 335, 339 (7th Cir. 1983).
[20] See Dep’t of Health & Human Servs., U.S. Food & Drug Admin., 21 CFR Part 310, Weight Control Drug Products for Over-the-Counter Human Use; Certain Active Ingredients; Final Rule, 56 Fed. Reg. 37791 (Aug. 8, 1991).
[21] Dietary Supplement Health and Education Act of 1994 (DSHEA). Pub. L. No. 103-417, 108 Stat. 4325 (1994).
[22] DSHEA permits structure function claims for dietary supplements provided that certain criteria are satisfied, including that the claims are substantiated and that the labeling includes a disclosure that the claim has not been evaluated by the FDA. FDCA, § 403(r)(6), 21 U.S.C. § 343(r)(6) (2025).
[23] FDCA § 403(r)(6)(B), 21 U.S.C. § 343(r)(6)(B) (1925).
[24] See U.S. Food & Drug Admin., Guidance for Industry: Substantiation for Dietary Supplement Claims Made Under Section 403r(6) of the Federal Food, Drug, and Cosmetic Act (Jan. 2009). Retrieved from https://www.fda.gov/regulatory-information/search-fda-guidance-documents/guidance-industry-substantiation-dietary-supplement-claims-made-under-section-403r-6-federal-food.
[25] Likely, the lack of enforcement is due to the lack of resources and the necessary investigative tools. For example, the FTC has the authority to force a company to produce the company’s substantiation for weight loss claims. In FDA’s case, such production would be voluntary.
[26] The Federal Trade Commission Act (FTC Act), § 5(a), 15 U.S.C. § 45(a) (2025).
[27] FTC Act, § 12, 15 U.S.C. § 12 (2025).
[28] A material claim is any claim that is likely to affect the consumer’s conduct or decision regarding a product or service. FTC Policy Statement on Deception, appended to Cliffdale Associates, Inc., 103 F.T.C. 110, 174 (1984).
[29] FTC v. Roca Labs, Inc., 345 F. Supp. 3d 1375, 1386 (M.D. Fla. 2018).
[30] Porter & Dietsch, Inc., 90 F.T.C. 770, 873 (1977), modified, 605 F.2d 294 (7th Cir. 1979), modified, 95 F.T.C. 806 (1980). See FTC v. Teami, LLC., No. 8:20-cv-518-T-33TGW (M.D. Fla. Mar. 17, 2020) (stipulated order).
[31] See Federal Trade Commission, Health Products Compliance Guidance (2022), p. 12 (“As a general matter, substantiation of health-related benefits will need to be in the form of randomized, controlled human clinical testing to meet the competent and reliable scientific standard.”). Retrieved from https://www.ftc.gov/business-guidance/resources/health-products-compliance-guidance.
[32] See Federal Trade Commission, Notice of Penalty Offenses Concerning Substantiation of Product Claims (2023). Retrieved from https://www.ftc.gov/system/files/ftc_gov/pdf/Substantiaton-NPO.pdf; FTC v. National Urological Group, Inc., 2017 U.S. Dist. Lexis 182256, 95-106 (2017).
[33] See Health Products Compliance Guidance, p. 11 (setting forth factors considered in determining appropriate level of substantiation).
[34] See, e.g., U.S. v. Jason Pharmaceuticals, Inc., No. Civ. 1:12-cv-01476 (D. D.C. Sept. 17, 2012) (Appendix A). Retrieved from https://www.ftc.gov/sites/default/files/documents/cases/2012/09/120910jasonpharmdecree.pdf.
[35] AMG Capital Mgmt., LLC v. FTC, 593 U.S. ___, 141 S. Ct. 1341 (2021).
[36] See, e.g., FTC v. Direct Mktg. Concepts, Inc., 624 F. 3d 1, 14 (1st Cir. 2010) (defendant’s gross receipts are an appropriate measure of damages); FTC v. Slim America, Inc., 77 F. Supp. 2d 1263, 1275 (S.D. Fla. 1999) (Section 13(b) permits consumer redress.).
[37] See FTC Act, §§ 5(m), 19, 15 U.S.C. §§ 45(m), 57b(b) (2025).
[38] See FTC v. Teami, LLC, No. 8:20-cv-00518 (M.D. Fla. 2020). Retrieved from https://www.ftc.gov/legal-library/browse/cases-proceedings/182-3174-teami-llc.
[39] Press Release, Federal Trade Commission, Penalty Offenses Concerning Weight Loss (2022). Retrieved from https://www.ftc.gov/enforcement/penalty-offenses/weightloss.
[40] Federal Trade Commission, Synopsis of Federal Trade Commission Determinations Concerning Promotion of Products and Plans for Weight Control (1983). Retrieved from https://www.ftc.gov/enforcement/penalty-offenses/weightloss.
[41] See FTC Act § 5(m)(1)(B), 15 U.S.C. §45(m)(1)(B).
[42] Press Release, Federal Trade Commission, FTC Publishes Inflation-Adjusted Civil Penalty Amounts for 2025 (2025). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2025/02/ftc-publishes-inflation-adjusted-civil-penalty-amounts-2025.
[43] Press Release, Federal Trade Commission, Anthony Calls for More Rigorous Self-Regulation (2002). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2002/04/anthony-calls-more-rigorous-self-regulation; Press Release, Federal Trade Commission, FTC Chairman Urges Media to Help Combat Deceptive Weight Loss Advertising (2003). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2003/02/ftc-chairman-urges-media-help-combat-deceptive-weight-loss-advertising.
[44] POM Wonderful LLC, 155 F.T.C. 1, 55 (2013), affirmed in part, POM Wonderful LLC v. FTC, 777 F.3d 478 (D.C. Cir. 2015).
[45] Porter and Dietch, supra note 9.
[46] Id. at 865.
[47] FTC v. NPB Advert., Inc., 218 F. Supp. 3d 1352, 1357 (M.D. Fla. 2016).
[48] See https://pubmed.ncbi.nlm.nih.gov/22291473/.
[49] Onakpoya, I., Terry, R., & Ernst, E. (2011). The use of green coffee extract as a weight loss supplement: A systematic review and meta-analysis of randomized clinical trials. Gastroenterology Research and Practice, 2011, 382852. https://doi.org/10.1155/2011/382852
[50] FTC v. NPB Advert., Inc., 218 F. Supp. 3d 1352, 1357 (M.D. Fla. 2016).
[51] Press Release, Federal Trade Commission, FTC Announces “Operation Waistline”—a Law Enforcement and Consumer Education Effort Designed to Stop Misleading Weight Loss Claims (1997). Retrieved from https://www.ftc.gov/news-events/news/press-releases/1997/03/ftc-announces-operation-waistline-law-enforcement-consumer-education-effort-designed-stop-misleading.
[52] Id.
[53] Press Release, Federal Trade Commission, FTC Seeks to Halt 10 Operators of Fake News Sites from Making Deceptive Claims About Acai Berry Weight Loss Products (2011). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2011/04/ftc-seeks-halt-10-operators-fake-news-sites-making-deceptive-claims-about-acai-berry-weight-loss.
[54] Press Release, Federal Trade Commission, Sensa and Three Other Marketers of Fad Weight-Loss Products Settle FTC Charges in Crackdown on Deceptive Advertising (2014). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2014/01/sensa-three-other-marketers-fad-weight-loss-products-settle-ftc-charges-crackdown-deceptive.
[55] Complaint at 5, FTC v. Sensa Products, LLC, No. 1:14-cv-00072 (E.D. ILL. Jan. 7, 2014). Retrieved from https://www.ftc.gov/sites/default/files/documents/cases/140107sensacmpt.pdf.
[56] Press Release, Federal Trade Commission, United States, Mexico, Canada (MUCH) Combat Weight Loss Fraud (2005). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2005/10/united-states-mexico-canada-much-combat-weight-loss-fraud.
[57] See, e.g., Communications Decency Act of 1996, Section 230, 47 U.S.C. § 230. (1996). Under most circumstances, Section 230 protects websites from liability for third-party content published on their websites.
[58] See Cent. Hudson Gas & Elec. Corp. v. Pub. Serv. Comm’n, 447 U.S. 557, 566 (1980).
[59] Federal Trade Commission, Weight Loss Advertising: An Analysis of Current Trends (2002). Retrieved from https://www.ftc.gov/reports/weight-loss-advertisingan-analysis-current-trends.
[60] Id. at 30.
[61] Id. at 24.
[62] Press Release, Federal Trade Commission, FTC to Hold Weight Loss Advertising Workshop (2002). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2002/11/ftc-hold-weight-loss-advertising-workshop.
[63] Federal Trade Commission, Deception in Weight-Loss Advertising Workshop: Seizing Opportunities and Building Partnerships to Stop Weight-Loss Fraud, A Federal Trade Commission Staff Report (Dec. 2003). Retrieved from https://www.ftc.gov/reports/deception-weight-loss-advertising-workshop-seizing-opportunities-building-partnerships-stop-weight.
[64] Remarks Before the Cable Television Advertising Bureau, FTC Chairman Timothy J. Muris (Feb. 11, 2003). Retrieved from https://www.ftc.gov/news-events/news/speeches/do-right-thing-apologies-spike-lee.
[65] Press Release, Federal Trade Commission, FTC Releases Guidance to Media on False Weight-Loss Claims Staff Report on Weight-Loss Advertising Workshop Made Available (2003). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2003/12/ftc-releases-guidance-media-false-weight-loss-claims.
[66] Federal Trade Commission, Gut Check: A Reference Guide for Media on Spotting False Weight Loss Claims (2014). Retrieved from https://www.ftc.gov/business-guidance/resources/gut-check-reference-guide-media-spotting-false-weight-loss-claims.
[67] Press Release, Federal Trade Commission, FTC Launches Big Fat Lie Initiative Targeting Bogus Weight-Loss Claims (2004). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2004/11/ftc-launches-big-fat-lie-initiative-targeting-bogus-weight-loss-claims.
[68] Press Release, Federal Trade Commission, FTC Releases Results of Weight-Loss Advertising Survey: While Media Screening of Fraudulent Ads Seems to Be Working, More Work is Needed, FTC Reports (2005). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2005/04/ftc-releases-result-weight-loss-advertising-survey.
[69] Federal Trade Commission, 2004 Weight-Loss Advertising Survey: A Report From the Staff of the Federal Trade Commission, p. 7–8 (Apr. 2005). Retrieved from https://www.ftc.gov/news-events/news/press-releases/2005/04/ftc-releases-result-weight-loss-advertising-survey.
[70] Id. at 8.
[71] Press Release, Federal Trade Commission, FTC Conference Explores How Government, Industry and Consumer Groups Can Work Together to Provide Consumers the Information They Need to Evaluate Weight Loss Products and Programs (1997). Retrieved from https://www.ftc.gov/news-events/news/press-releases/1997/10/ftc-conference-explores-how-government-industry-consumer-groups-can-work-together-provide-consumers.
[72] Press Release, Federal Trade Commission, “Partnership for Healthy Weight Management” to Release Voluntary Consumer Disclosure Guidelines (1999). Retrieved from https://www.ftc.gov/news-events/news/press-releases/1999/02/partnership-healthy-weight-management-release-voluntary-consumer-disclosure-guidelines.
FDLI is a nonprofit membership organization that offers education, training, publications, and professional networking opportunities in the field of food and drug law. As a neutral convener, FDLI provides a venue for stakeholders to inform innovative public policy, law, and regulation. Articles and any other material published in Update represent the opinions of the author(s) and should not be construed to reflect the opinions of FDLI, its staff, or its members. The factual accuracy of all statements in the articles and other materials is the sole responsibility of the authors.
Recent Update Magazine Articles

 Mr. CLELAND is presently Of Counsel with Arnall Golden Gregory LLP. He retired from the Federal Trade Commission in 2022 after a thirty-year career with the agency, twenty of those as Assistant Director of the Division of Advertising Practices.
Mr. CLELAND is presently Of Counsel with Arnall Golden Gregory LLP. He retired from the Federal Trade Commission in 2022 after a thirty-year career with the agency, twenty of those as Assistant Director of the Division of Advertising Practices.






